Abstract
Objective:Although advances in endoscopic techniques, resection of complex lesions that occupying multiple compartments in the skull base still represents significant challenges, since surgical outcomes may be compromised by insufficient exposure and inappropriate techniques. The question of whether to perform endoscopic surgery or to use open craniofacial approach remains unresolved. Nevertheless, careful assessment of lesion characteristics must be carried out in selection of the appropriate trajectory.
Methods:Between May 2006 and November 2016, 16 patients experienced resection of extensive lesions in the skull base via maxillary swing approach. Data regarding clinical findings and technical considerations as shown in case illustrations were discussed.
Results:Complete resection was achieved in all patients. Pathological findings were diverse, while the majority were schwannomas (10 cases, 62.5%) followed by meningiomas (3 cases, 18.75%). The complications were managed as shown in case illustrations, and symptoms improved with time. The follow-up duration ranged from 49 to 177 months (median, 102 months), while 2 patients were lost. There was no postoperative mortality, and 1 patient who experienced a relapse of meningioma 72 months after surgery is still under observation due to asymptomatic status.
Conclusions:Our preliminary results suggest that the maxillary swing approach can be an alternative option in management of extensive, recurrent, and hypervascularized masses with fibrous or calcified consistency in the skull base, which may represent challenges for the endoscopic procedure. Future studies are required to demonstrate the surgical indications and efficiency.
Keywords:Maxillary swing approach; Skull base pathology; Technical considerations; Lesion characteristics
Introduction
The extreme complexity of the skull base is associated with challenging regional anatomy and variable pathologies of lesions. Neoplasms arising from the paranasal sinuses, orbits, pterygopalatine fossa (PPF), infratemporal fossa (ITF), sellar or clivus may exhibit transdural growth pattern [1-4]. Moreover, a number of subdural masses can spread to the craniofacial region, exhibiting a more aggressive biological behavior. Technically, extirpation of lesions extending towards multiple compartments remains formidable because of proximity to vital neurovascular structures without adequate exposure [3, 5].
Controversy remains regarding the optimal surgical procedure for extensive lesions in the skull base [2, 3]. The maxillary swing approach has gained wide access in case of large tumors and obtained a complete resection in one-stage [3, 6-9]. However, since implementation of endoscopic techniques, surgeons would argue strongly against open surgery for tumors residing in the cranial fossa due to postoperative morbidity and unacceptable cosmetic results [6, 10, 11] Endoscopic surgery also provided data on series with excellent outcomes even when piecemeal resection was performed [11-15].
The characteristics of lesion is a major decision-making element of surgical approach. Full exposure of deep-seated extensive or recurrent tumors which distort the anatomical landmarks cannot be easily achieved through a narrow corridor [1, 8, 16] Surgical struggle to dissect lesions with significant fibrosis or calcification within the restrictive surgical cavity poses great risks to the vital neurovascular structures [3, 11]. Likewise, piecemeal resection may cause severe intraoperative bleeding from dissection planes of hypervascularized lesion remnant [11]. Therefore, the maxillary swing approach may be feasible under special circumstances.
Notably, indications of this approach have yet to be fully described in the endoscopic era, leaving the optimal treatment paradigm unresolved. In the present study, 16 patients with lesions occupying multiple compartments in the skull base underwent open surgery, and clinical data accompanied by technical considerations based on our experience as shown in case illustrations were reviewed and discussed.
Materials and Methods
Patient populationThe study was approved by the ethical committee of the National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College. Informed consent was obtained from patients in our cohort.
Between May 2006 and November 2016, 16 patients suffering extensive lesions in the skull base underwent surgical treatment via the maxillary swing approach in our department. The clinical findings were summarized in Table 1. In this cohort, previous treatment was performed in 7 patients (43.75%) who experienced local relapse before admission to our hospital.
All surgeries were performed by a senior neurosurgical team with >10 years of experience in skull base surgery, including > 300 cases of open skull base surgery and endoscopic skull base surgery prior to the study period.
Table 1. Demographics, clinical, and pathologic characteristics
| Variables | Value |
|---|---|
| No. of eligible patients | 16 |
| Mean age, range (years) | 48.5, 29-67 |
| Gender, No. (%) | |
| Male | 8 (50) |
| Female | 8 (50) |
| Presenting symptoms, No. (%) | |
| Headache | 6 (37.5) |
| Limitation of extracollar movements | 6 (37.5) |
| Facial numbness/pain | 5 (31.25) |
| Visual defects | 5 (31.25) |
| Anosmia | 5 (31.25) |
| Nasal stenosis/obstruction | 4 (25) |
| Hear impairment | 2 (12.5) |
| Facial deformity | 2 (12.5) |
| Exophthalmos | 1 (6.25) |
| Facial palsy | 1 (6.25) |
| Local relapse before admission to our hospital, No. (%) | 7 (43.75) |
| Previous treatment, No. (%) | |
| Transcranial surgery | 4 (25) |
| Endoscopic surgery + radiotherapy | 2 (12.5) |
| Endoscopic surgery + transcranial surgery | 1 (6.25) |
| Mean maximum diameter, range (cm) | 6.69, 3.6-10.5 |
| Extension across the midline, No. (%) | 6 (37.5) |
| Transdual growth pattern, No. (%) | 6 (37.5) |
| CS invasion, No. (%) | 9 (56.25) |
| Complete resection rate (%) | 100 |
| Pathology, No. (%) | |
| Schwamoma | 10 (62.5) |
| Meningioma | 3 (18.75) |
| MPNST (WHO III.) | 1 (6.25) |
| Fibrous dysplasia | 1 (6.25) |
| Simple cyst | 1 (6.25) |
| Complications, No. (%) | |
| Facial numbness/pain | 4 (25) |
| Dry eye | 3 (18.75) |
| CSF leakage | 2 (12.5) |
| Palatal fistula | 2 (12.5) |
| Visual defects | 1 (6.25) |
| Triismus | 1 (6.25) |
| Epistaxis | 1 (6.25) |
| Intracranial infection | 1 (6.25) |
| Lost to follow-up, No. (%) | 2 (12.5) |
| Median follow-up duration, range (month) | 102.49-177 |
| Mortality (%) | 0 |
| Recurrence rate (%) | 7.14 (1/14) |
The maximum diameter of lesions ranged from 3.6 to 10.5cm (mean, 6.69 cm), while extension across the midline was found in 6 cases (37.5%). Notably, violation of the cavernous sinus (CS) was present in 9 cases (56.25%), and 6 patients (37.5%) harbored lesions occupying the subdural area. The lesions mostly exhibited aggressive features of bone destruction together with gross calcification on computed tomography (CT) scans (9 cases, 56.25%), and vivid contrast enhancement was seen on T1-weighted post-gadolinium magnetic resonance imaging (MRI) in 13 patients (81.25%).
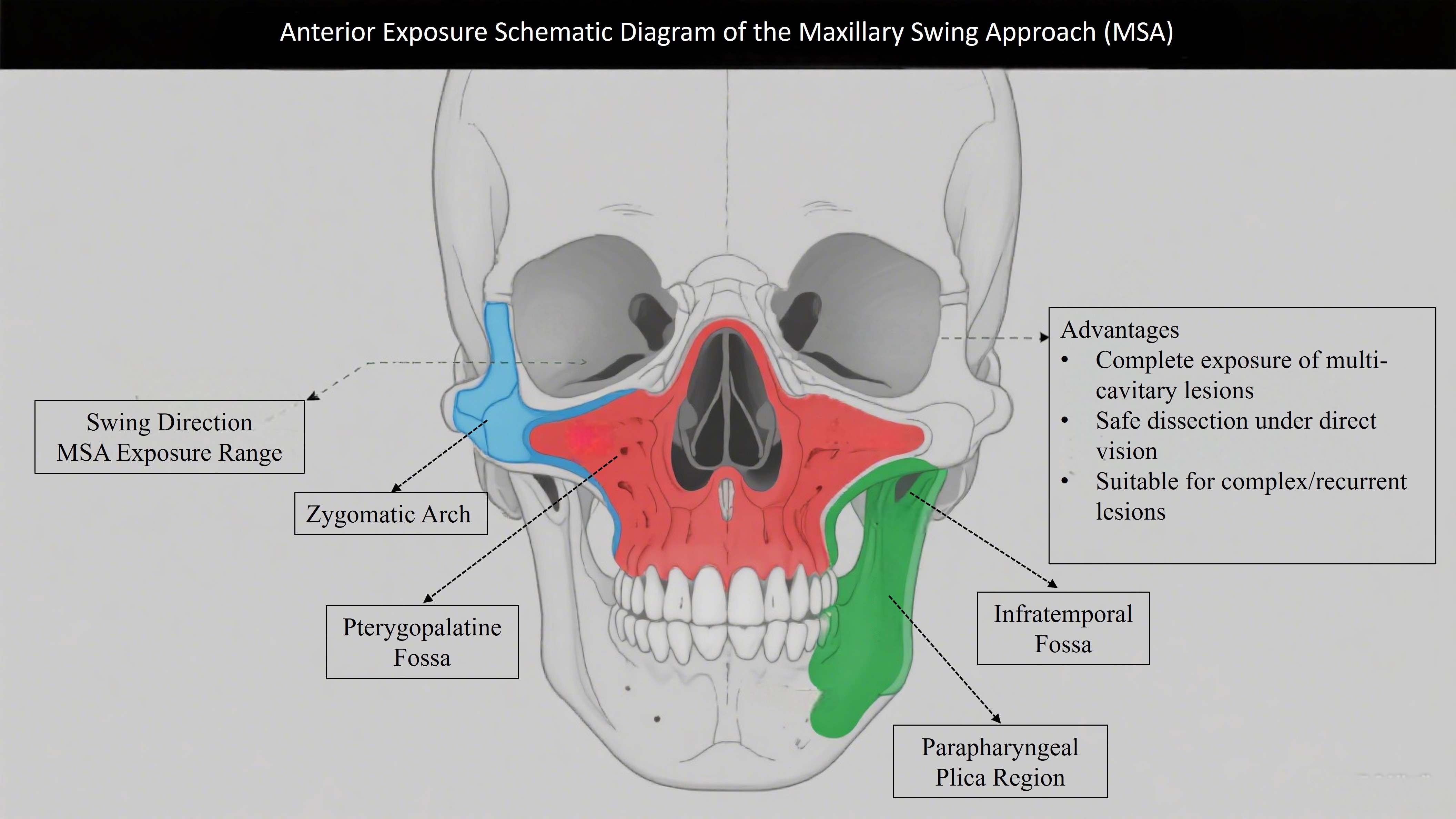
Surgical procedureAfter administration of general anesthesia, the patient was placed supine. Then the maxillary swing approach was performed using the standard procedure [2]. In general, a Weber-Ferguson incision was given followed by osteotomies to separate the maxilla from the zygomatic arch and pterygoid plates. Thereafter, the hard palate was divided in the center, favoring mobilization and reflection of the entire maxilla laterally. The nasopharynx, oropharynx, parapharyngeal space, fossa of Rosenmüller, PPF, ITF, sellar, and clivus could be widely exposed under direct visualization. Removal of the middle turbinate and posterior end of the nasal septum including the vomer displayed the contralateral compartments. Then extirpation of large extensive lesion was achieved using standard microsurgical techniques after expanding surgical corridor bilaterally and posteriorly. Eventually, multilayer reconstruction of the skull base was carried out followed by repositioning of the maxillary osteocutaneous unit.
Surgical approach selection criteriaThe decision to adopt the maxillary swing approach (MSA) was based on comprehensive evaluation of lesion characteristics and technical feasibility of alternative approaches. Specifically, MSA was preferred over endoscopic or other open approaches if the lesions met one or more of the following criteria: (1) Imaging features: maximum diameter >5 cm, cross-midline extension, cavernous sinus invasion, obvious calcification or fibrosis on CT/MRI; (2) Clinical features: recurrent lesions after previous surgery, hypervascularity on angiography, or firm consistency (fibrous/calcified) predicted to complicate piecemeal resection; (3) Technical limitations of endoscopy: inability to achieve sufficient exposure of multi-compartment involvement (e.g., simultaneous involvement of infratemporal fossa, pterygopalatine fossa, and clivus) or risk of uncontrollable bleeding during endoscopic dissection. These criteria were established through multidisciplinary team discussions (neurosurgery, otolaryngology, and radiology) and were consistent throughout the study period, rather than surgeon-dependent.
Results
Surgical aspects and pathological findingsMicroscopically total resection was achieved in all patients. The complications noted in our study were as mentioned in Table 1 and managed as shown in case illustrations. The most common tissue diagnosis was schwannoma (10 cases, 62.5%) followed by meningioma (3 cases, 18.75%).
Follow-upThe median follow-up duration was 102 months (range, 49 to 177 months), but 2 patients were lost. These 2 patients were confirmed to have no residual tumor or severe complications at the 12-month postoperative visit before being lost to follow-up due to relocation. There was no mortality, while 1 patient who had meningioma experienced locoregional tumor recurrence 72 months after surgery, giving a recurrence rate of 7.14% (1/14). Observation and monitoring were recommended because of asymptomatic status.
Case illustrationsCase 1
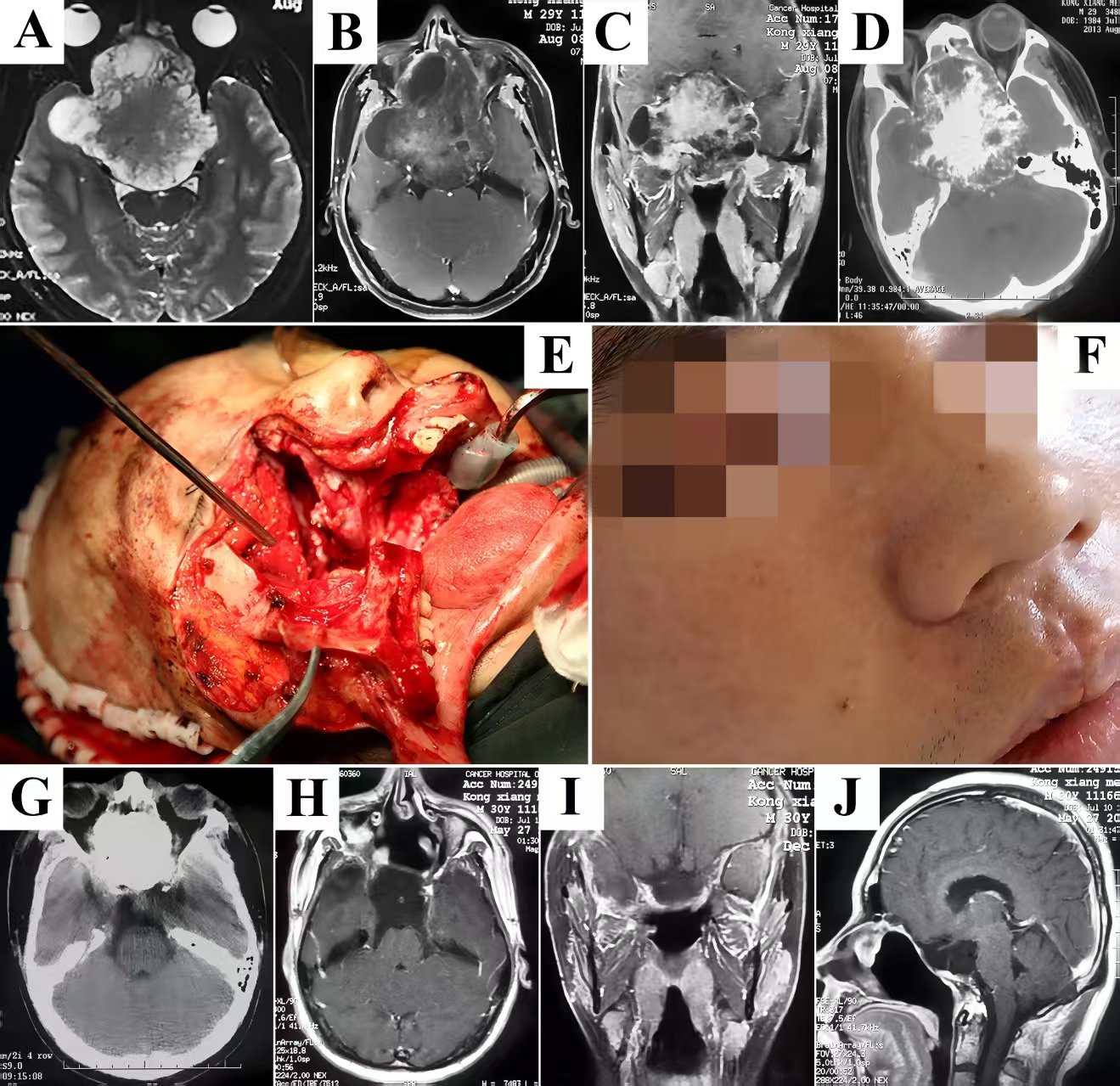
A 29-year-old man initially presented with nasal obstruction and anosmia. Then he complained of blurring 8 months later, and was referred to our clinic because of precipitate bitemporal hemianopsia and severe exophthalmos. Preoperative T2-weighted MRI showed a large lesion with heterogeneous signal intensity in the skull base (Fig. 1A). Contrast enhancement was uneven, which depicted the cystic/necrotic areas within the hypervascularized mass growing inferiorly into the nasal sinuses, anteriorly into the orbits, posteriorly to the sellar, clivus, and petrous apex, bilaterally into the CS, and infiltrating the temporal lobes (Fig. 1B&C). Considering the difficulties in resection of the extensive and calcified lesion in a bloodless view and reconstruction of the skull base through a narrow corridor under endoscopic visualization, the modified maxillary swing approach was selected by expanding nasal osteotomy to the contralateral side. The bony-hard lesion was visualized and dissected along its capsule surface, struggling to keep the displaced and stretched cranial nerves within the CS intact. Pure circumferential stripping in a single piece along the brain-lesion interface was accomplished followed by reflection of frontotemporal scalp flap to harvest malleable autologous tissues for reconstruction (Fig. 1E). Postoperative CT scans confirmed complete removal of the lesion without intracranial hematoma or brain contusion (Fig. 1G). The diagnosis of fibrous dysplasia was finally confirmed by pathological analysis. The postoperative course was uneventful and the patient was discharged with improvement in visual function. He was followed up annually, and no recurrence was seen on contrast T1-weighted MRI (Fig. 1H-J).
Figure 1. Representative case of fibrous dysplasia harboring transdural growth pattern. Heterogeneous signal intensity on plain T2-weighted MRI in axial plane demonstrated an extensive lesion containing cystic/necrotic areas (A). Significantly uneven enhancement seen on T1-weighted post-gadolinium MRI in axial (B) and coronal (C) planes confirmed the hypervascularized lesion infiltrated the brain parenchyma. There was stone-like calcification in the core of the mass which also presented aggressive features of bone destruction on CT scans (D). After adequate exposure of retromaxillary area, complete excision of the lesion was accomplished followed by multilayer reconstruction of the skull base to prevent CSF leakage and obliterate surgical cavity using the temporalis fascia, pericranium, and muscle flap (E). The cosmetic results were acceptable without visible facial scars 3 months after surgery (F). Postoperative CT scans (G) and contrast T1-weighted MRI (H-J) exhibited the excellent outcomes. MRI, magnetic resonance imaging; CT, computed tomography. CSF, cerebrospinal fluid.
Case 2
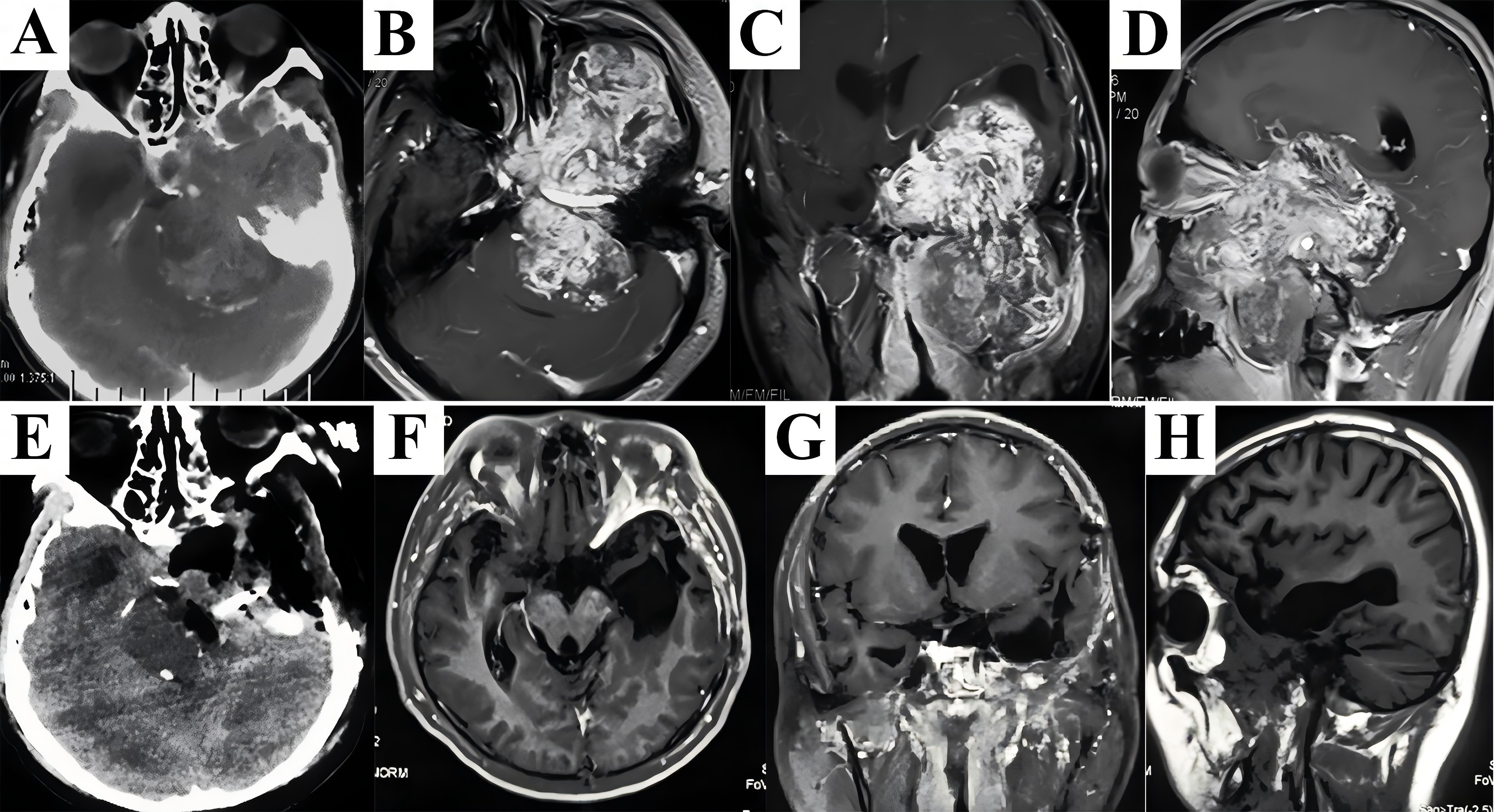
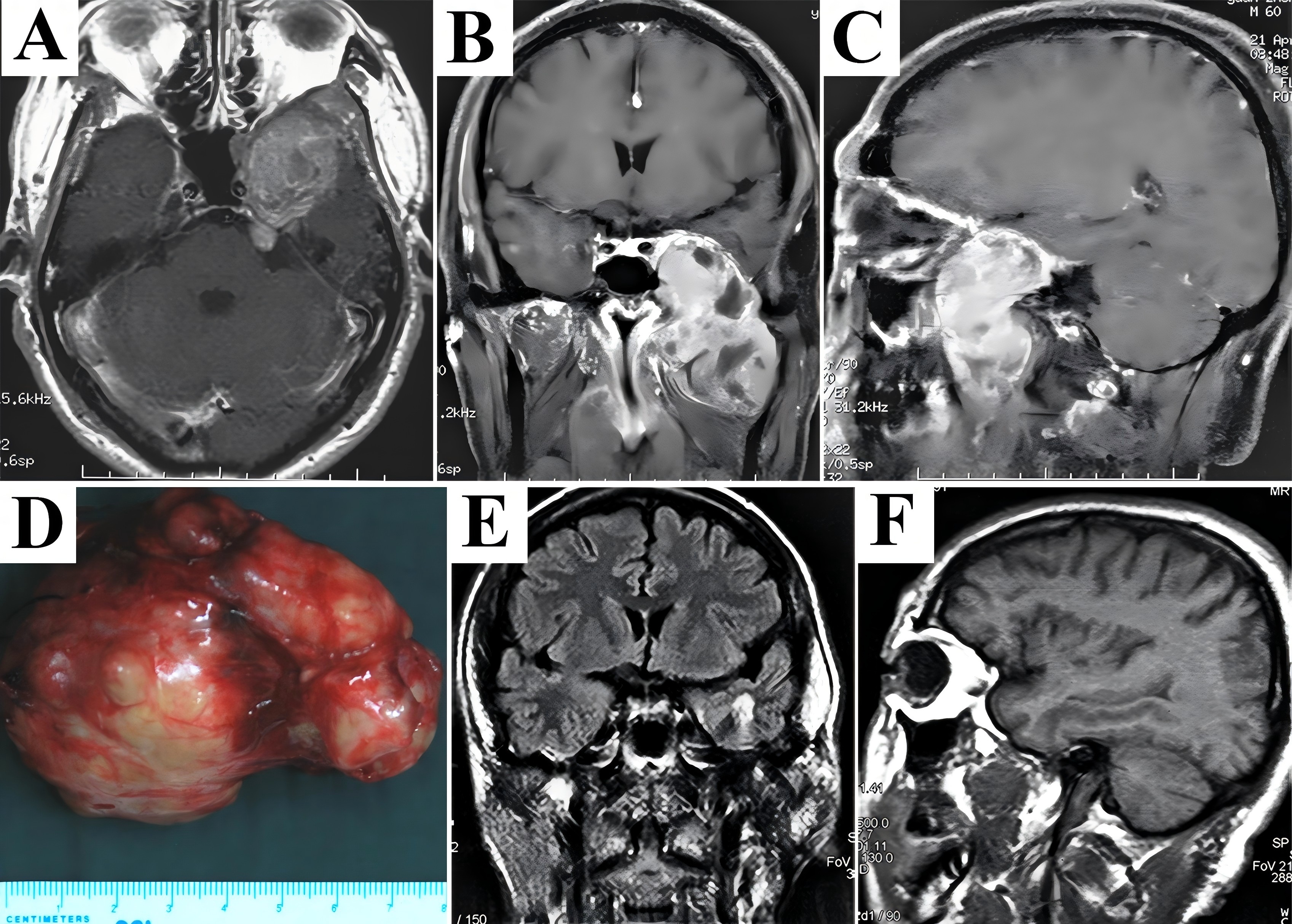
A 32-year-old man tolerated nasal obstruction, anosmia, and persistent facial numbness for 4 months, and sought medical attention when facial deformity was developed. Flattening of the left nasolabial fold and slightly mouth drooping were observed on admission. Preoperative CT scans showed a huge lesion centered at the left petroclival region destructing the petrous bone and occupying multiple craniofacial compartments (Fig. 2A). Significant enhancement of the extensive mass abutting the mandible and migrating into the subdural region on MRI (Fig. 2B-D) presented great challenges since sufficient exposure and complete resection in one-stage without severe bleeding could not be achieved solely from any approach. Therefore, the maxillary swing approach combined with the Kawase’s approach were performed. After reflection of the maxilla laterally, the lesion was exposed and feeding arteries (probably the internal maxillary artery) were ligated and transected. The extracranial portion of the rubber-like multilobulated tumor was transected at its extension into the Meckel’s cave using a scalpel because of fibrous consistency. Dissection continued through the petroclival region to the cerebellopontine angle (CPA) using the Kawase’s approach, and remnant subdural segment was completely stripped away from the arachnoid membrane, allowing resolution of the mass effect on the brainstem. Following hemostasis and antibiotic irrigation of surgical field, multilayer reconstruction of skull base started with dura suture. Then the extradural area was packed with the temporalis fascia, periosteum, and pedicled temporalis muscle to reinforce dural repair. CT scans immediately after surgery demonstrated complete resection (Fig. 2E). The patient developed facial numbness and suffered temporary dry eye postoperatively. Intracranial infection secondary to cerebrospinal fluid (CSF) leak occurred 4 days after surgery, and was resolved by antibiotic therapy accompanied by lumbar drain. The final diagnosis based on histological findings was schwannoma. During follow-up period of 49 months, there was no evidence of recurrence (Fig. 2F-H).
Figure 2. A combined craniofacial resection of the giant and rubbery schwannoma was performed. Preoperative CT scans revealed an extensive lesion destructed the petrous bone and migrated both anteriorly and posteriorly (A). Significant enhancement mixed with discrete hypointensity on contrast T1-weighted MRI in axial (B), coronal (C), and sagittal (D) planes probably demonstrated a hypervascularized lesion with features of fibrosis. After removal of the extracranial portion via maxillary swing approach, dissection proceeded along the intradural segment through the Kawase’s approach to obtain a complete resection in one-stage, which was shown on CT scans immediately after surgery (E). Postoperative MRI detected no tumor recurrence during the follow-up period (F-H). CT, computed tomography; MRI, magnetic resonance imaging.
Case 3
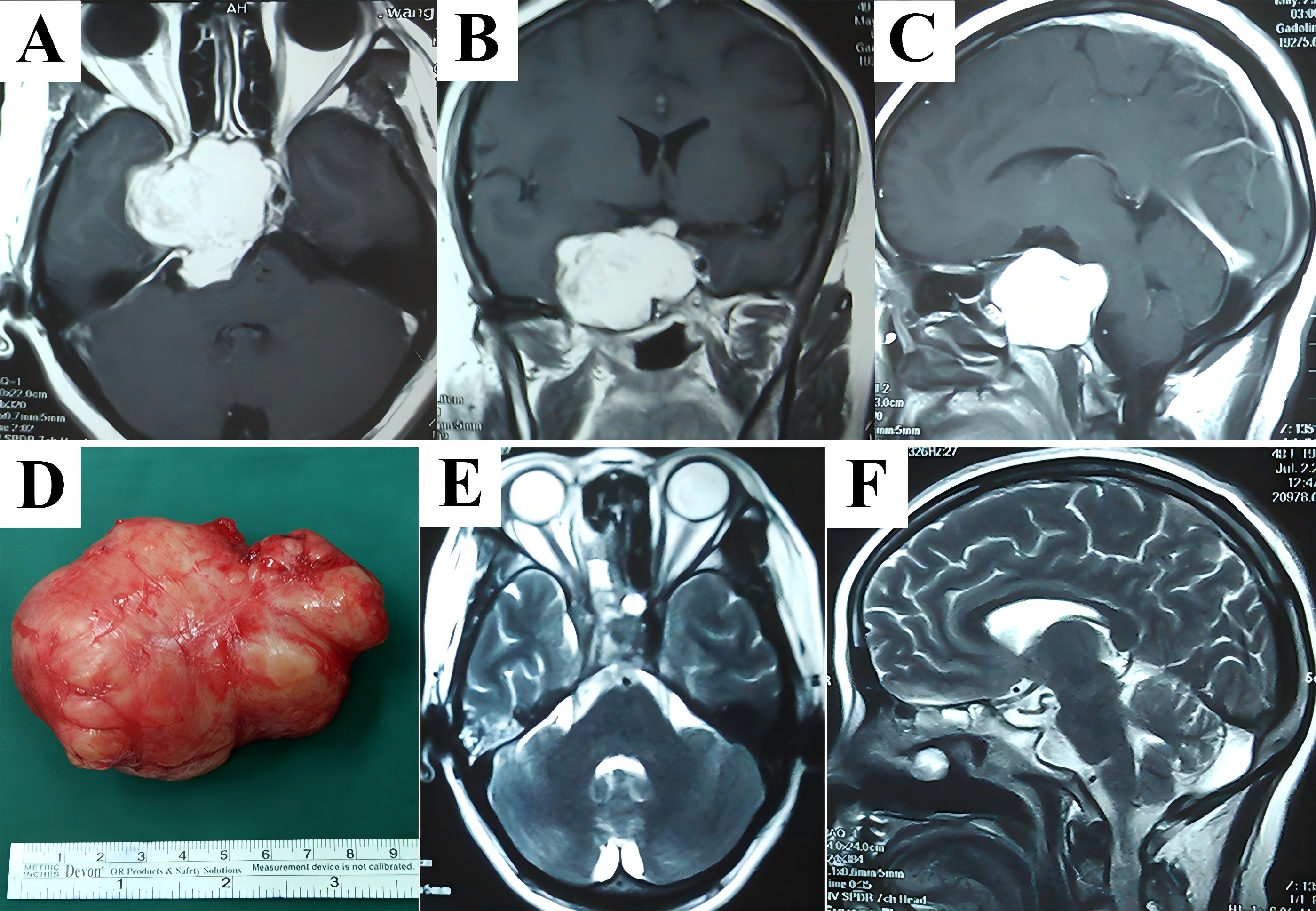
A 49-year-old woman suffered 3-year history of progressive headache, blurring, diplopia, and ptosis. On neurological examination, vision loss, partial cranial nerve (CN) III palsy, and complete CN VI palsy of the right eye were observed. T1-weighted post-gadolinium MRI revealed significantly homogeneous contrast enhancement of a lesion which involved the sellar, CS, clivus, parasellar area, and petroclival region (Fig. 3A-C). The endoscopic endonasal technique or extradural subtemporal transpetrosal access (Kawase’s approach) was not recommended because severe bleeding due to tumor debulking and dissection across the cranial nerves might pose great risks to the patient. Standard maxillary swing procedure was carried out. After identifying superoposterior displacement of the cranial nerves, the tumor was peeled off along its capsule as a whole under an almost bloodless view (Fig. 3D). There was no surgical complication, and the tissue diagnosis was schwannoma. The patient was relieved of visual defects and extraocular movements limitation at follow-up, and MRI scans demonstrated no evidence of tumor growth (Fig. 3E&F).
Figure 3. A hypervascularized schwannoma migrating across the midline to the contralateral side was totally resected as a whole. Vivid contrast enhancement was shown in T1-weighted MRI in axial (A), coronal (B), and sagittal (C) planes. The lesion was peeled off along its capsule surface in a single piece under an almost bloodless view. Histopathological examination of excised specimen (D) determined the diagnosis of schwannoma, and postoperative T2-weighted MRI in axial (E) and sagittal (F) planes demonstrated an absence of tumor. MRI, magnetic resonance imaging.
Case 4
A 59-year-old man received transcranial surgery for skull base schwannoma 5 years ago. Although pathological examination found tumor exhibiting characteristics of active proliferation, he did not undergo postoperative radiotherapy. The major complaints for which he sought medical help included blurring, ptosis, limitation of extraocular movements. An extensive tumor occupying the parasellar area, Meckel’s cave, temporal fossa, PPF, ITF, and parapharyngeal space was depicted in contrast T1-weighted MRI (Fig. 4A-C). The maxillary swing approach was selected, which could provide enhanced lateral exposure of the tumor that predominantly allocated within extracranial space. The recurrent tumor firmly adhered to the adjacent tissues and challenged the dissection planes. Under direct visualization, not only stripping of tumor from lateral wall of the CS in a single piece but confirmation of intact dura and negative margins by intraoperative fresh-frozen sectioning were performed to obtain local disease control (Fig. 4D). Postoperative MRI showed total resection of the tumor (Fig. 4E&F). The patient developed trismus and palatal fistula after surgery. He wore the obturator dental plate to facilitate swallowing, and the majority of the fistulae shrunk with time accompanied by improvement of the trismus. The patient who had a definitive histological diagnosis of the malignant peripheral nerve sheath tumor (MPNST) (WHO Ⅲ) refused to receive adjuvant radiotherapy. Fortunately, no locoregional tumor recurrence was observed after a period of 129 months.
Figure 4. Recurrent MPNST (WHO Ⅲ) allocated in the skull base were completely resected via maxillary swing approach. A large and extensive tumor occupying multiple compartments including the parasellar area, Meckel’s cave, temporal fossa, PPF, ITF, and parapharyngeal space as depicted in axial (A), coronal (B), and sagittal (C) contrast T1-weighted MRI exhibited heterogeneous enhancement due to necrosis or cystic degeneration. The patient experienced tumor resection in a piecemeal fashion before admission to our hospital. In contrast, following reflection of the maxilla, we identified and removed the recurrent tumor as a whole (D). Postoperative MRI confirmed total removal of the tumor (E, F). MPNST, malignant peripheral nerve sheath tumor; PPF, pterygopalatine fossa; ITF, infratemporal fossa; MRI, magnetic resonance imaging.
Discussion
The skull base represented a complex intersection between the sellar, clivus, petroclival region, orbits, PPF, ITF, paranasal sinuses, nasopharynx, etc [4, 17]. Extensive lesions may wholly occupy several compartments or even cross the midline towards the contralateral side in communication with subdural space, leading to the surgical frustration and therapeutic dilemma [10, 11]. Therefore, surgical approach is indispensable to provide excellent exposure of particular locations from various angles and allow for direct visualization of dissection planes [10].
The skull base lesions occupying multiple compartments have been reached by several open surgical techniques, such as Fisch pre-auricular infratemporal approach and Derome transbasal approach [18, 19]. Recently, there has been a trend away from traditional approaches towards endoscopic procedure which may improve visualization and achieve minimal invasion. The endoscopic techniques appear to have progressively supplanted open surgery for benign tumors involving the cranial fossa [6, 11]. Although piecemeal resection under improved endoscopic visualization can provide excellent outcomes [8, 14, 15], open surgery seems comparable in special circumstances including i) huge extensive masses occupying multiple compartments, ii) hypervascularized lesions exhibiting features of significant fibrosis or stone-like calcification, and iii) recurrent tumors firmly adhering to the adjacent tissues and distorting the anatomical landmarks.
First successfully performed by Joseph Gensoul in 1827, the maxillectomy inspired Irwin Moore who practiced the lateral rhinotomy almost a century later, paving the way for addressing central skull base lesions [20]. In 1986, Hernández pioneered an access to the clivus and retromaxillary areas by temporary disarticulation of the maxilla attached to the cheek, which was then named maxillary swing approach [7, 21, 22]. The indications were expanded by Wei et al. who provided a sufficient exposure down to the level of the parapharyngeal space [7, 22]. The study period (2006-2016) coincided with rapid advances in endoscopic skull base techniques. During this period, our team gradually integrated endoscopic evaluation into preoperative planning: after 2012, endoscopic exploration was routinely performed for potential candidates, and MSA was reserved for lesions that failed endoscopic feasibility assessment (e.g., extensive calcification, firm adhesion to neurovascular structures, or multi-compartment involvement beyond endoscopic reach). This dynamic adjustment ensured that MSA was selected only when endoscopic approaches were deemed inadequate. More recently, in the endoscopic era, maxillary swing approach still enabled sufficient visual field and working space by performing osteotomies at the pterygoid plates, pterygoid process, zygomatic arch, and nasal septum, with possibilities of further identification of the contralateral compartments [3, 10, 21-24]. Comparative analysis with endoscopic outcomes for similar complex lesions further supports the niche role of MSA. Previous studies on endoscopic approaches for multi-compartment skull base lesions have reported gross total resection rates of 65-80% for large (>5 cm) or recurrent tumors, with higher complication rates (e.g., CSF leak in 15-25% and residual tumor in 10-20%) [11,14]. In contrast, our series achieved 100% gross total resection with manageable complications (e.g., CSF leak in 1 case, 6.25%), which may be attributed to MSA's advantage in direct visualization and circumferential dissection of hypervascular or fibrotic lesions. However, it should be noted that endoscopic approaches remain superior for small to medium-sized, non-calcified, and midline lesions with minimal extracranial extension, highlighting the complementary role of MSA in complex scenarios.
In the present study, we adopted the maxillary swing approach to address extensive lesions in the skull base. Based on our retrospective data, we consider that maxillary swing approach can be an alternative option to endoscopic surgery in some circumstances. (i) Adequate visualization for extirpation of extensive lesions - Surgical freedom and angle of attack are determined by the soft tissue and bony structures which impede the maneuverability of instruments [1, 25]. Through a more restrictive corridor, reaching deeper and more lateral lesions which grow into multiple compartments as illustrated in Case 1 and 2 may be more difficult using the endoscopic techniques because surgical procedure is limited by the nares, nasolacrimal duct, and bony walls of paranasal sinuses [8, 25, 26]. In our series, the maxillary swing procedure has provided enhanced exposure as a result of lateral entry on the horizontal plane and direct anteroposterior angle of attack in the sagittal section. The line of vision on surgical cavity was straight in almost all cases. Furthermore, significantly augmented working space was achieved, decreasing surgical struggle caused by conflict between frustrated angled instrumentation. (ii) Circumferential stripping of hypervascularized masses with fibrous or calcified consistency - It has to be emphasized that, the choice of surgical approach should be partially determined by the characteristics of lesions [13]. Circumferential dissection of hypervascularized and hard mass as a whole is feasible since severe bleeding cannot be avoided effectively when performing piecemeal resection and few surgical instruments can provide fragmented debulking as shown in Case 2 and 3. Therefore, a widened approach should be used to facilitate devascularization and insure safe dissection of neurovascular structures [12-15]. In our study, the maxillary swing approach provided direct access to the fibrous or calcified lesions and sufficient margins around them which were then peeled off in a single piece, resulting in minimal blood loss. (iii) Identifying dissection planes of recurrent tumors - The anatomical landmarks of the cranial fossa have always been distorted after local relapse, particularly when patients experienced adjuvant therapy [13]. The recurrent tumors that are firmly adherent to the surrounding structures have challenged the dissection planes and represented an obstacle to the endoscopic procedure [12]. Following swing of the maxilla, we established a spacious access to identify tumor interface and detached the planes of adherence using sharp dissection, obtaining tumors eradication without dividing them. The negative surgical margins were confirmed by intraoperative fresh-frozen sectioning in case of malignant tumor (see Case 4).
Previously, the maxillary swing approach has been associated with unacceptable aesthetic sequelae caused by prominent postoperative facial scars [8, 14, 16, 27]. The early embryological development of the face is accomplished by merging and fusion of the frontonasal process, maxillary processes, medial nasal processes, lateral nasal processes, and mandibular processes [28]. The merging and fusion sites such as the philtrum, sides and alae of the nose, and lateral canthus have a shortage of dermis [26, 28]. The dermal injuries have been identified as contributing to scar formation. Hence, these merging and fusion sites are potential regions where surgical incisions can be made with decreased likelihood of facial scars formation [8, 10, 26, 28]. In our series, we planned the Weber-Ferguson incision, and the cosmetic outcomes were acceptable since almost no visible facial scars could be found 3 months after surgery. Despite its advantages, MSA has inherent limitations: (1) Compared with endoscopic approaches, it is associated with longer operation time (mean 380 min in our series) and hospital stay (mean 14 days vs. 7-10 days for endoscopy); (2) Potential complications include trismus (1 case, 6.25%) and palatal fistula (1 case, 6.25%), which require prolonged rehabilitation; (3) It is technically demanding, requiring expertise in craniofacial reconstruction to avoid skull base defects. These limitations emphasize that MSA should be considered as a complementary rather than universal approach.
Although the maxillary swing approach is recommended for large extensive lesions allocated in the skull base, perioperative complications and prolonged recovery time call the principles of minimally invasive surgery in question particularly in the endoscopic era. In our retrospective study, patient selection bias may exist particularly in the beginning of the series. With regard to diverse pathologies of lesions, it is difficult to draw direct comparison of prognostic factors between different cohorts due to a limited number of patients. Future prospective randomized controlled trials comparing outcomes would better characterize this technique and demonstrate the surgical indications.
Conclusion
We have provided case illustrations and reviewed clinical data regarding the maxillary swing approach for skull base lesions. The preliminary results suggest that this open procedure can be used to supplement endoscopic surgery in management of huge, extensive, and hypervascularized masses with fibrous or calcified consistency. Sufficient exposure leads to complete excision in a single piece with acceptable morbidity and fair cosmetic results. Patients harboring recurrent tumors also benefit from this procedure.
Declarations
Authors' Contributions
JW and HQ designed the study and revised the manuscript. HL and HC drafted the manuscript. QL and CF reviewed and revised the manuscript. All authors made substantial contributions to the study and approved the submitted version.
Acknowledgements
The authors also wish to thank Meng-Yan Zhang for technical assistance.
Funding information
This study was supported by the CAMS Innovation Fund for Medical Sciences (2022-I2M-C&T-B-063), National Natural Science Foundation of China (No. 82472722) and Beijing Hope Run Special Fund of Cancer Foundation of China (No. LC2022B18).
Ethics Approval and Consent to Participate
All procedures performed in studies involving human participants were con-ducted in accordance with the ethical standards of the institutional and/or national research committee and with the 1975 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all the patients included in the study.
Competing Interests
The authors declare that there is no conflict of interest regard-ing the publication of this article.
Statements and Declarations
The authors declare that there is no conflict of interest regarding the publication of this article.
Data availability
Not Applicable.
References
 Figures
Figures References
References Peer
Peer Information
InformationFigure 1. Representative case of fibrous dysplasia harboring transdural growth pattern. Heterogeneous signal intensity on plain T2-weighted MRI in axial plane demonstrated an extensive lesion containing cystic/necrotic areas (A). Significantly uneven enhancement seen on T1-weighted post-gadolinium MRI in axial (B) and coronal (C) planes confirmed the hypervascularized lesion infiltrated the brain parenchyma. There was stone-like calcification in the core of the mass which also presented aggressive features of bone destruction on CT scans (D). After adequate exposure of retromaxillary area, complete excision of the lesion was accomplished followed by multilayer reconstruction of the skull base to prevent CSF leakage and obliterate surgical cavity using the temporalis fascia, pericranium, and muscle flap (E). The cosmetic results were acceptable without visible facial scars 3 months after surgery (F). Postoperative CT scans (G) and contrast T1-weighted MRI (H-J) exhibited the excellent outcomes. MRI, magnetic resonance imaging; CT, computed tomography. CSF, cerebrospinal fluid.

Figure 2. A combined craniofacial resection of the giant and rubbery schwannoma was performed. Preoperative CT scans revealed an extensive lesion destructed the petrous bone and migrated both anteriorly and posteriorly (A). Significant enhancement mixed with discrete hypointensity on contrast T1-weighted MRI in axial (B), coronal (C), and sagittal (D) planes probably demonstrated a hypervascularized lesion with features of fibrosis. After removal of the extracranial portion via maxillary swing approach, dissection proceeded along the intradural segment through the Kawase’s approach to obtain a complete resection in one-stage, which was shown on CT scans immediately after surgery (E). Postoperative MRI detected no tumor recurrence during the follow-up period (F-H). CT, computed tomography; MRI, magnetic resonance imaging.

Figure 3. A hypervascularized schwannoma migrating across the midline to the contralateral side was totally resected as a whole. Vivid contrast enhancement was shown in T1-weighted MRI in axial (A), coronal (B), and sagittal (C) planes. The lesion was peeled off along its capsule surface in a single piece under an almost bloodless view. Histopathological examination of excised specimen (D) determined the diagnosis of schwannoma, and postoperative T2-weighted MRI in axial (E) and sagittal (F) planes demonstrated an absence of tumor. MRI, magnetic resonance imaging.

Figure 4. Recurrent MPNST (WHO Ⅲ) allocated in the skull base were completely resected via maxillary swing approach. A large and extensive tumor occupying multiple compartments including the parasellar area, Meckel’s cave, temporal fossa, PPF, ITF, and parapharyngeal space as depicted in axial (A), coronal (B), and sagittal (C) contrast T1-weighted MRI exhibited heterogeneous enhancement due to necrosis or cystic degeneration. The patient experienced tumor resection in a piecemeal fashion before admission to our hospital. In contrast, following reflection of the maxilla, we identified and removed the recurrent tumor as a whole (D). Postoperative MRI confirmed total removal of the tumor (E, F). MPNST, malignant peripheral nerve sheath tumor; PPF, pterygopalatine fossa; ITF, infratemporal fossa; MRI, magnetic resonance imaging.

Peer-review Terminology
Identity transparency: Single anonymized
Reviewer interacts with: Editor
Details
This is an open access article under the terms of the Creative Commons Attribution License(http://creativecommons.org/licenses/by/4.0/), which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Publication History
Received 2025-06-18
Accepted 2025-07-27
Published 2025-08-10


